Cholescintigraphic evaluation of gallbladder contraction in correlation with serum thyrotropin and serum cholesterol
Apollo Hospital, Chennai, India
Author and article information
Cite this as
Abdul Rahim S, Indirani M, Shelley S (2023) Cholescintigraphic evaluation of gallbladder contraction in correlation with serum thyrotropin and serum cholesterol. Open J Hepatol. 2023; 5(1): 1-5. Available from: 10.17352/ojh.000009
Copyright License
© 2023 Abdul Rahim S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background and aims: To study the effect of serum thyrotropin (Sr.TSH) and serum cholesterol on gallbladder contraction by cholescintigraphy.
Methods: This is a prospective cross-sectional study comprising 77 patients for evaluation of gallbladder function in patients with biliary pain. All of them underwent 99mTc- Mebrofenin fatty meal cholescintigraphy following a standard institutional protocol apart from regular investigations. Dynamic images were acquired for 90 minutes with a standardised fatty meal given at the 60th minute. Gallbladder emptying kinetics was determined with Gallbladder Ejection Fraction (GBEF) calculated at 30 minutes post-meal. Relevant statistical methods were applied for interpretation.
Results: The mean of GBEF was 60.82% ± 16.65% and the mean of Sr.TSH was 3.72 ± 3.88 mIU/ml, there was a negative correlation between GBEF and Sr.TSH (Pearson correlation coefficient r = -0.535, p < 0.05). The mean serum cholesterol was 180.61 ± 46.34 mg/dl, there was a negative correlation between GBEF and serum cholesterol (Pearson’s correlation coefficient r = -0.685, p < 0.05). The mean GBEF in euthyroid and hypothyroid patients were 65.04% ± 15.04% and 47.27% ± 15.93% respectively (P < 0.05).
Conclusion: Hypothyroidism is one of the key factors affecting gallbladder contractility and should not be overlooked in the management of functional gallbladder disorders. Cholescintigraphy can accurately assess gallbladder function qualitatively and quantitatively, and it can help (aid) physicians in diagnosing and decision-making on the management of biliary pain in hypothyroidism.
Functional Gallbladder Disorder (FGBD) or Gallbladder (GB) dyskinesia [1] is a controversial disorder having a constellation of symptoms, mainly biliary pain [2,3]. It was also known by various names in the past like chronic acalculous gallbladder dysfunction, acalculous biliary disease, chronic acalculous cholecystitis, and biliary dyskinesia. Clinically biliary pain is described as a right upper quadrant or epigastric pain that manifests as slow, steady pain, which lasts at least for 30 minutes and is severe enough to interrupt daily activities or require consultation with a physician [4]. Diagnosis of FGBD is a consideration in patients with biliary pain without gallstone or gall bladder sludge, functional nature of gallbladder disorder is established by the absence of hepato-pancreatic morphologic and biochemical abnormality [5] (Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST), bilirubin, alkaline phosphatase, amylase, and lipase).
Decreased gall bladder emptying on cholescintigraphy is the only objective characteristic of functional gallbladder disorder, and it is diagnosed by low Gallbladder Ejection Fraction (GBEF) on cholescintigraphy [6] Despite these clinical criteria there exists confusion and significant overlap with other Functional Gastrointestinal Disorders (FGIDs).
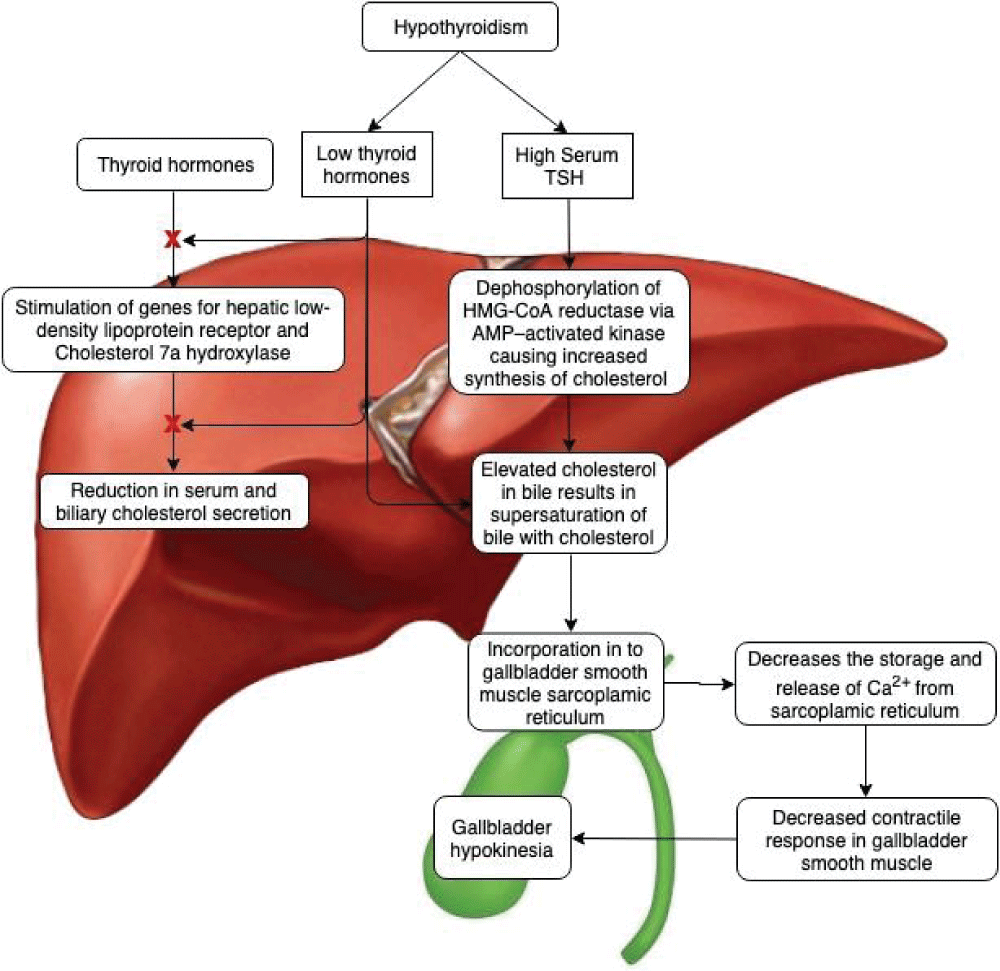
The pathophysiology of gallbladder hypokinesia in FGBD is not well understood, but it is hypothesised to develop in the presence of cholesterol supersaturation of bile [7,8,], which may interfere with Ca2+ storage and release causing reduced gallbladder smooth muscle contraction resulting in hypokinesia. Cholesterol supersaturation of bile is established in many etiologies, and hypothyroidism is one of them [9]. Animal studies have indicated that the flow of bile into the duodenum is decreased in a hypothyroid state. This could be due to the composition of bile, dysmotility of the gallbladder, and dysfunction of the sphincter of Oddi [10,11]. There are various studies that indicate an abnormality in gallbladder function and the sphincter of Oddi pressure are independent of each other [12,13].
The effect of hypothyroidism on the sphincter of Oddi function has been well documented, and its pathophysiology through Thyroid Hormone Receptors (TR) β1 and β2 [9] receptors are known. However, there was no significant documented literature on the effect of hypothyroidism on gallbladder function by cholescintigraphy.
Methods
Prospective cross-sectional study, for a period of 1 Year 11 months. The study was approved by the Institutional Ethics Committee and written informed consent was obtained from all the patients.
A detailed history was taken, and the Rome IV questionnaire for functional biliary disorders was used. All the subjects were screened with Ultrasonography (USG) of the upper abdomen and upper gastrointestinal endoscopy to rule out organic causes of disease. Thyroid function tests, fasting blood sugar, and serum cholesterol were evaluated. Liver and pancreatic function tests were confirmed to be normal.
Selection of subjects
Subjects aged 18 years and above with normal and abnormal thyroid function tests were included in the study consecutively by convenient sampling till the sample size was reached.
Exclusion criteria
- Structural gallbladder disease that would explain the symptoms (e.g., GB polyp, Adenomyomatosis).
- Diabetes Mellitus and cholelithiasis.
- Patients on hyperthyroid or hypothyroid medication
- Cholecystectomy and congenital absent gallbladder.
- Pregnant or lactating females.
Methodology
- With prior informed consent all anti-motility drugs, laxatives, calcium channel blockers, cholinergic, anticholinergic, and neuroleptic medications were stopped two days prior to the scheduled scan.
- Fasting duration of at least 6 hours and not more than 12 hours was ensured.
- 99mTc-Mebrofenin was administered intravenously.
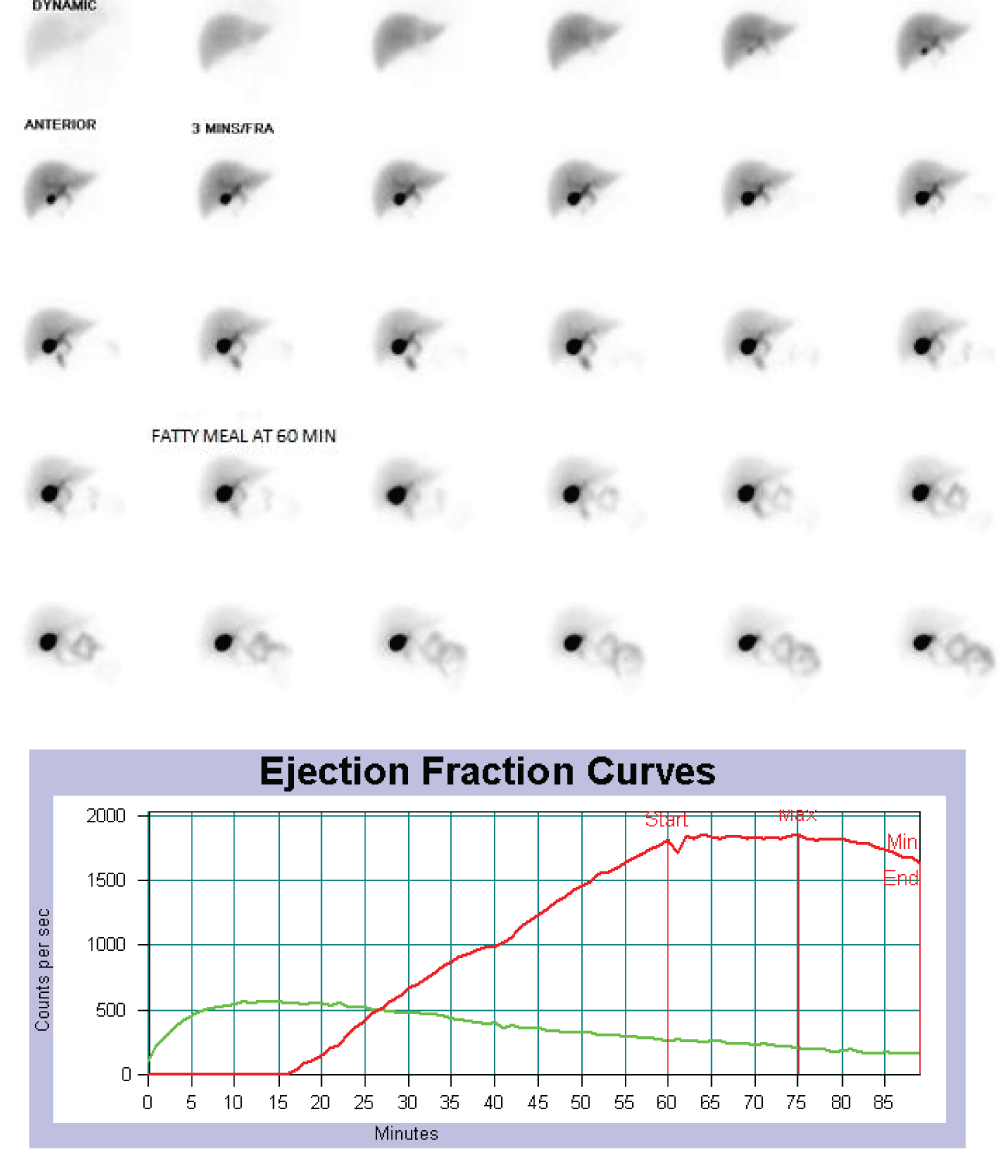
- Dynamic images of a 1-minute duration were taken over a period of 90 minutes.
- A standardized fatty blend with approximately 488 Kcal containing 25 g of fat was given at the 60th minute as a cholecystagogue to provoke gallbladder contraction. (Fatty meal preparation: Add 25 g of butter in 50 mL of hot milk and mix it thoroughly. Then add 150 mL of yogurt and 1 g of salt and whip in a blender [14]).
Image processing and interpretation
Computer-generated Regions of Interest (ROI) were drawn around the gallbladder and background on hepatic parenchyma, gallbladder ejection fraction was calculated by using the formula.
Grading was done by (In house grading at our institution after standardisation) [15]. GBEF > 60% - Normal, < 60% - Hypokinetic, Filling after the fatty meal –Dyskinetic Liver function tests, pancreatic function tests, and bilirubin were not included in the study.
Patients were categorised into groups based on Sr.TSH.
- Euthyroid – Sr. TSH 0.42-4.2 mIU/ml – 51 patients.
- Hypothyroid – Sr. TSH > 4.2 mUI/ml – 22 patients.
- Hyperthyroid – Sr. TSH < 0.42 mIU/ml – 3 patients.
Statistical methods
The Pearson correlation coefficient was computed for the association between categorical bivariate continuous variables (Namely GBEF with Sr.TSH and GBEF with Sr.FT4). Student T-test was used to compare continuous data (i.e. GBEF) between two groups (namely Euthyroid, and Hypothyroid).
Ethical guidelines
Ethical clearance was obtained from the institutional ethics committee before starting the study. Standard operating procedures of the nuclear medicine department were followed.
Results
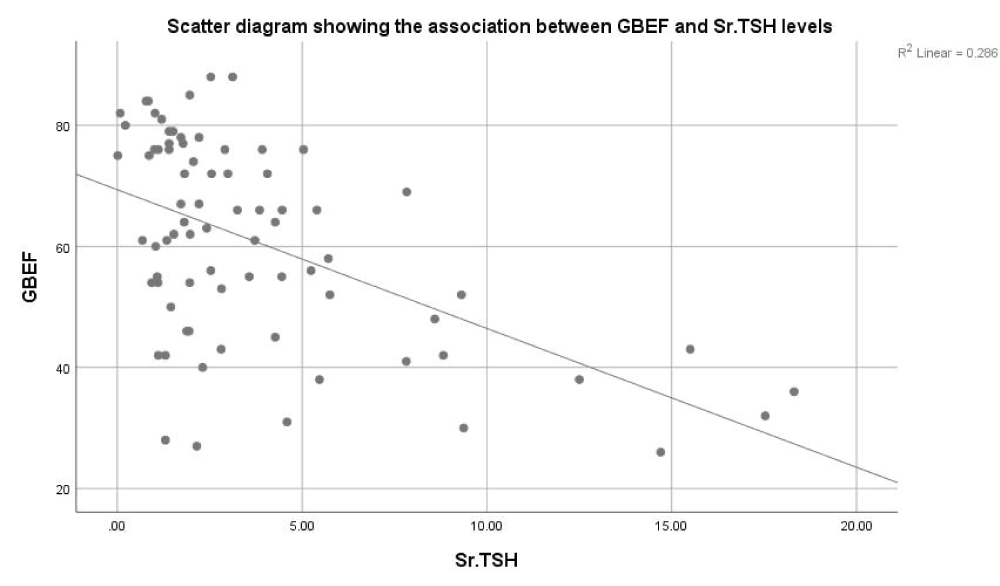
The mean of GBEF was 60.82% ± 16.65% and the mean of Sr.TSH was 3.72 ± 3.88 mIU/ml, It was found that as serum TSH increased, the GBEF reduced, and it was concluded that there was a statistically significant negative correlation between GBEF and Sr. TSH values (Pearson correlation coefficient r = -0.535, p < 0.05) (Figure 1).
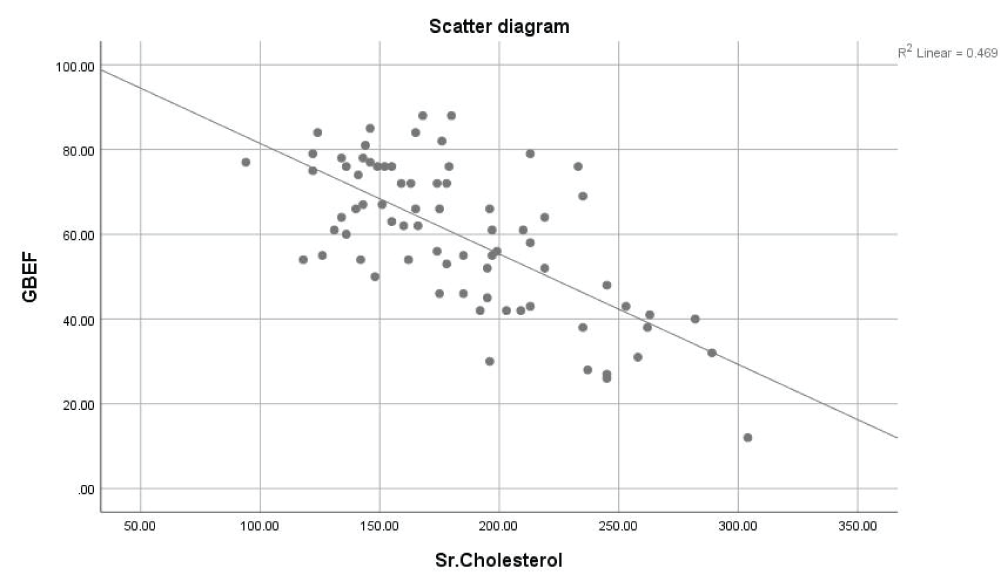
The mean Sr. Cholesterol was 180.61 ± 46.34 mg/dl. There was a statistically significant negative correlation between GBEF and Sr. Cholesterol values (Pearson’s correlation coefficient r = -0.685, p < 0.05), i.e., as serum cholesterol increased, the GBEF showed a decline (Figure 2).
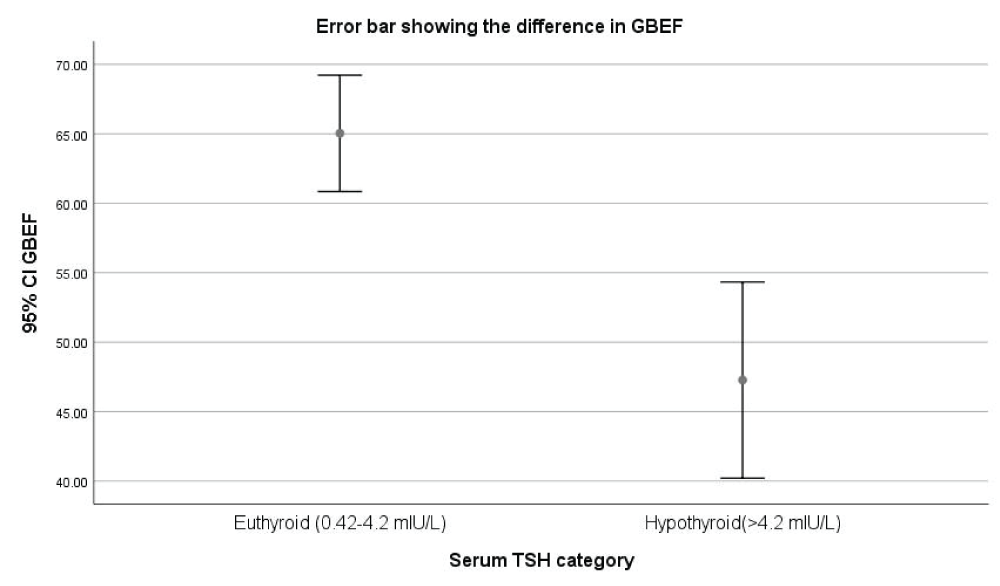
The mean GBEF in euthyroid patients and hypothyroid patients were 65.04% ± 15.04% and 47.27% ± 15.93% respectively, and the difference in mean was statistically significant (P < 0.05) (Figure 3).
Discussion
In laboratory studies, human gallbladder muscle strips exposed to bile with excess cholesterol had shown a marked reduction in the force of spontaneous phasic contractions, impairment of gallbladder motility may result from cholesterol affecting one or more steps of excitation-contraction coupling and cannot be explained by receptor-agonist interaction. It is speculated that excess cholesterol in extracellular space could cause stiffening of cholesterol-rich membranes. Excess of cholesterol in cholesterol-poor membranes like the endoplasmic reticulum reduces calcium storage and release [15]. Studies on ground squirrels concluded that the primary contractile defect in cholesterol gallstone disease does not reside in intracellular signal transduction pathways but involves sarcoplasmic membrane by decreasing the Ca2+ storage and release from it, this ends in the waning of contractile response in gallbladder smooth muscle [16].
Raised serum cholesterol might cause bile to supersaturate with cholesterol [9]. Studies on male Sprague–Dawley rats demonstrated thyroid hormones stimulate genes responsible for hepatic low-density lipoprotein (LDL) receptor and Cholesterol 7α hydroxylase, and this results in enhanced removal of LDL from serum [17]. Studies on human subjects have described, that in a state of hypothyroidism, there is decreased clearance of cholesterol from serum, causing an increase in serum cholesterol concentration [17]. An increase in serum and biliary cholesterol in hypothyroidism is not just mediated by low thyroid hormones, but also by TSH mediated mechanism. Studies on genetically thyroid-stimulating hormone receptor (Tshr) -deficient mice have shown that TSH causes the dephosphorylation of HMG-CoA reductase via AMP-activated kinase, causing increased cholesterol synthesis [17]. In our study, there was a significant negative correlation between Sr. cholesterol and GBEF (Pearson correlation coefficient r = - 0.685, p < 0.05) (Figure 4).
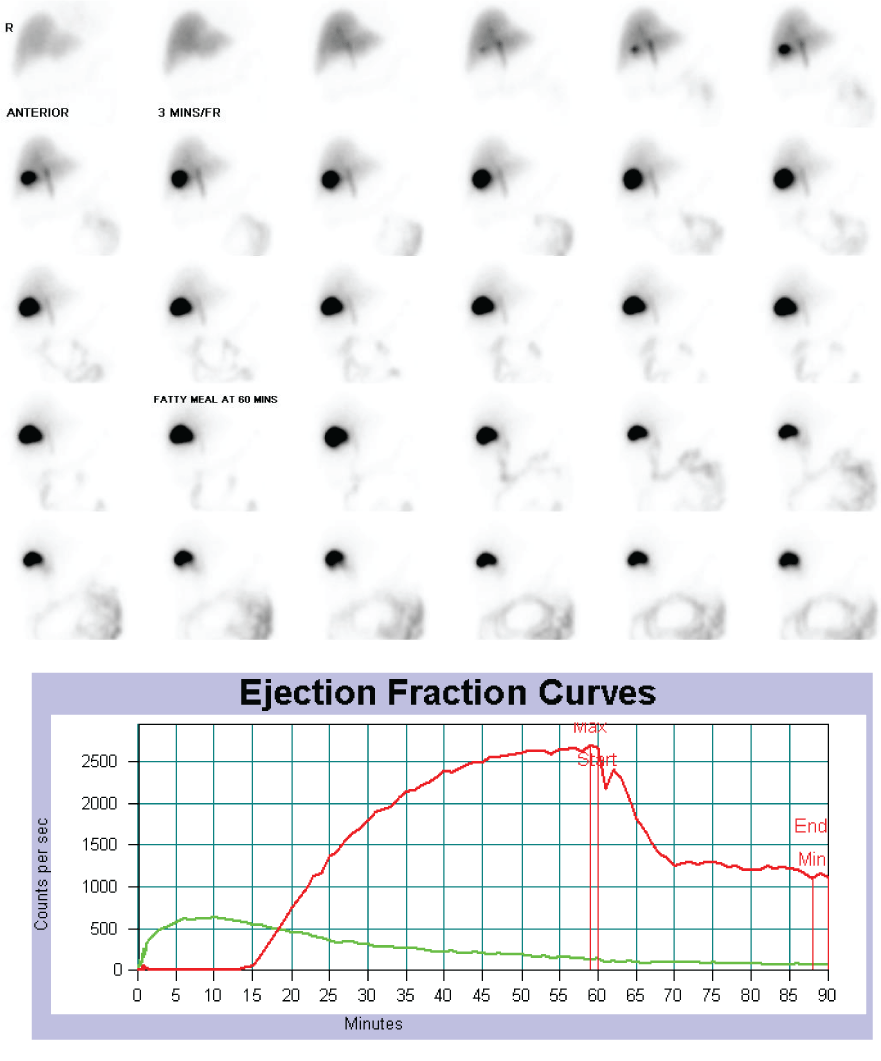
Management of biliary pain in functional gallbladder disorder has been a therapeutic dilemma, and the status of cholecystectomy in them remains controversial [18]. In our study, we found that hypothyroidism does have an effect on gallbladder contraction, causing gallbladder dyskinesia (Figures 5,6). If this subset of patients with biliary pain and low GBEF were given thyroxin replacement as a part of management before cholecystectomy, unwanted cholecystectomy could be avoided if there is a clinical improvement. However, a larger prospective multicentric trial with a standardised protocol for cholescintigraphy is needed to validate our findings. (Sincalide is not available in India so standardisation of fatty meals is a difficult part to achieve in every centre).
Study limitations
- Fatty meals used at our centre and GBEF calculated at 30 min post-meal were based on a study published by our centre. A multicentre analysis is needed to standardize the protocol.
- The number of patients who presented with biliary pain and hyperthyroidism was low to study the effect of hyperthyroidism on GBEF.
Conclusion
Hypothyroidism is one of the key factors affecting gallbladder contractility and should not be overlooked in the management of functional gallbladder disorders. Cholescintigraphy can accurately assess gallbladder function qualitatively and quantitatively, and it can help (aid) physicians in diagnosing and decision-making on the management of biliary pain in hypothyroidism.
- Joseph Elmunzer B, Elta GH. Biliary tract motor function and dysfunction. Mark Feldman, Lawrence S. Friedman, and Lawrence J. Brandt (eds). In: Sleisenger & Fordtran's gastrointestinal and liver disease, 10th ed. Philadelphia: WB Saunders; 2016; 1067-1076.
- Settembre C, D'Antonio E, Moscato P, Loi G, Santonicola A, Iovino P. Association among Disorders of Gut-Brain Interaction (DGBI) and Fibromyalgia: A Prospective Study. J Clin Med. 2022 Feb 3;11(3):809. doi: 10.3390/jcm11030809. PMID: 35160260; PMCID: PMC8836992.
- Drossman DA, Hasler WL. Rome IV-Functional GI Disorders: Disorders of Gut-Brain Interaction. Gastroenterology. 2016 May;150(6):1257-61. doi: 10.1053/j.gastro.2016.03.035. PMID: 27147121.
- Cotton PB, Elta GH, Carter CR, Pasricha PJ, Corazziari ES. Gallbladder and sphincter of Oddi disorders. Gastroenterology. 2016 May 1; 150(6): 1420-9.
- Hansel SL, DiBaise JK. Functional gallbladder disorder: gallbladder dyskinesia. Gastroenterol Clin North Am. 2010 Jun;39(2):369-79, x. doi: 10.1016/j.gtc.2010.02.002. PMID: 20478492.
- Corazziari E, Shaffer EA, Hogan WJ, Sherman S, Toouli J. Functional disorders of the biliary tract and pancreas. Gut. 1999 Sep;45 Suppl 2(Suppl 2):II48-54. doi: 10.1136/gut.45.2008.ii48. PMID: 10457045; PMCID: PMC1766688.
- Petersen BT. Functional Gall‐Bladder and Sphincter of Oddi Disorders. Practical Gastroenterology and Hepatology: Liver and Biliary Disease: Liver and Biliary Disease. 2010 Sep 17: 365-73.
- Amaral J, Xiao ZL, Chen Q, Yu P, Biancani P, Behar J. Gallbladder muscle dysfunction in patients with chronic acalculous disease. Gastroenterology. 2001 Feb;120(2):506-11. doi: 10.1053/gast.2001.21190. PMID: 11159891.
- Laukkarinen J, Sand J, Nordback I. The underlying mechanisms: how hypothyroidism affects the formation of common bile duct stones-a review. HPB Surg. 2012;2012:102825. doi: 10.1155/2012/102825. Epub 2012 Sep 19. PMID: 23049165; PMCID: PMC3459253.
- Laukkarinen J, Sand J, Aittomäki S, Pörsti I, Kööbi P, Kalliovalkama J, Silvennoinen O, Nordback I. Mechanism of the prorelaxing effect of thyroxine on the sphincter of Oddi. Scand J Gastroenterol. 2002 Jun;37(6):667-73. doi: 10.1080/00365520212492. PMID: 12126244.
- Kulkarni V, Ramteke H, Lamture Y, Gharde P. A Review of Synchronous Findings of Hypothyroidism and Cholelithiasis. Cureus. 2022 Oct 14;14(10).
- Ruffolo TA, Sherman S, Lehman GA, Hawes RH. Gallbladder ejection fraction and its relationship to sphincter of Oddi dysfunction. Dig Dis Sci. 1994 Feb;39(2):289-92. doi: 10.1007/BF02090199. PMID: 8313810
- Kalloo AN, Sostre S, Meyerrose GE, Pasricha PJ, Szabo Z. Gallbladder ejection fraction. Nondiagnostic for sphincter of Oddi dysfunction in patients with intact gallbladders. Clin Nucl Med. 1994 Aug;19(8):713-9. PMID: 7955753.
- Jain AS, Simon S, Muthukrishnan I, Mohan AT, Balsubramaniam R. Standardization of "In-House fatty meal" methodology for cholescintigraphy. Indian J Nucl Med. 2015 Oct-Dec;30(4):303-8. doi: 10.4103/0972-3919.164021. PMID: 26430312; PMCID: PMC4579613.
- Behar J, Lee KY, Thompson WR, Biancani P. Gallbladder contraction in patients with pigment and cholesterol stones. Gastroenterology. 1989 Dec;97(6):1479-84. doi: 10.1016/0016-5085(89)90392-2. PMID: 2583414.
- Xu QW, Shaffer EA. The potential site of impaired gallbladder contractility in an animal model of cholesterol gallstone disease. Gastroenterology. 1996 Jan;110(1):251-7. doi: 10.1053/gast.1996.v110.pm8536864. PMID: 8536864.
- Liu H, Peng D. Update on dyslipidemia in hypothyroidism: the mechanism of dyslipidemia in hypothyroidism. Endocr Connect. 2022 Feb 7;11(2):e210002. doi: 10.1530/EC-21-0002. PMID: 35015703; PMCID: PMC8859969.
- Xu KJ, Brock JC, Goussous N, Sill AM, Cunningham SC. Functional gallbladder disorder: Interim analysis of a prospective cohort study. Am J Surg. 2023 Dec 13:S0002-9610(23)00655-4. doi: 10.1016/j.amjsurg.2023.12.010. Epub ahead of print. PMID: 38110322.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
