Pathogenesis and complications of non-alcoholic steatohepatitis and its management by nutraceuticals
1Nutrition and Food Sciences Department, National Research Centre, Egypt
2A member of the National Nutritional Sciences Committee, Academy of Scientific Research and Technology, Egypt
Author and article information
Cite this as
Al-Okbi SY (2024) Pathogenesis and complications of non-alcoholic steatohepatitis and its management by nutraceuticals. Open J Hepatol. 2024; 6(1): 1-3. Available from: 10.17352/ojh.000010
Copyright License
© 2024 Al-Okbi SY. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Non-Alcoholic Steatohepatitis (NASH) is a real health problem that is commonly associated with obesity and diabetes. Cardiovascular diseases, liver carcinoma, and liver failure might develop from NASH if not well managed. The present article deals with the pathogenesis of non-alcoholic steatohepatitis and its complications in addition to exploring the importance of using nutraceuticals in its prevention and treatment.
Non-Alcoholic Fatty Liver Diseases (NAFLD) affect more than 20% of the world population. The spectrum of NAFLD includes simple steatosis, steatohepatitis, fibrosis, cirrhosis, hepatocellular carcinoma, and hepatic failure. Steatosis is a simple deposition of fat in the liver, steatosis does not represent a health problem however progression to Non-Alcoholic Steatohepatitis (NASH) would be the real health problem since NASH is a risk factor for cardiovascular diseases (CVDs) and on the other hand might be advanced to liver cirrhosis, hepatocellular carcinoma and might be ended by liver failure [1-4].
A sedentary lifestyle, overnutrition, inflammation, oxidative stress, insulin resistance, genetic factors, and lipotoxicity have a strong correlation with the incidence of NASH. Excessive consumption of fructose and saturated fat might exacerbate the induction of NASH. Fructose elevates lipid lipogenesis in the liver through the de novo pathway and produces a reduction of beta-fatty acids oxidation [2,5].
Metabolic syndrome includes a cluster of components comprising fatty liver, dyslipidemia, visceral obesity, glucose intolerance, insulin resistance, and hypertension [6]. Obese and diabetic subjects are more commonly suffering from steatosis than normal. Patients with NAFLD were demonstrated to have high glycosylated hemoglobin, obesity, elevated or normal Alanine Transaminase (ALT) and normal Aspartate Transaminase (AST) however in more advanced stages they might show AST/ALT to be 0.8 [7]. It is worth mentioning that elevations in ALT and AST are indicative of liver damage.
The pathophysiology of NASH is not well understood however insulin resistance has been reported to influence hepatic, muscle, and adipocytes metabolisms of fat and sugar which leads to deposition of lipids in the hepatocytes. Fat deposition by itself in the liver could lead to increased oxidative stress and inflammation. Leptin resistance due to malfunctioning of its receptor might also contribute to NASH development [8]. Reduced adiponectin that induces hepatic inflammation is commonly encountered in obese, NASH, and diabetics [9]. Therefore the role of adipokines, leptin and adiponectin, cannot be ignored in NASH progression which is mediated by elevated oxidative stress [2]. The imbalance of colonic microbiota has been proposed as one of the major causes of NASH due to transforming inflammation in the liver with further modulation of liver pathology by microbiota metabolite [10].
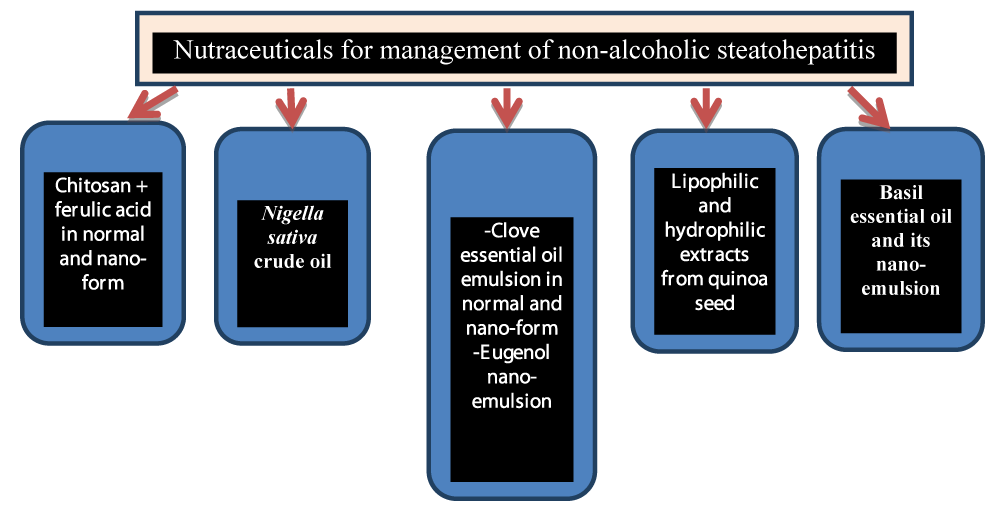
So far there is no efficient remedy for NASH [11,12] however insulin sensitizers like biguanides (metformin), lipid-lowering drugs like statins and other therapies such as urodeoxycholic acid and pentoxyfilline might be used [13]. Natural agents that could be beneficial are under experimental investigations. From the natural products; nutraceuticals are functional food ingredients purified from food and are considered safe natural products that could be efficient in mitigating steatohepatitis. There are no internationally agreed definitions of nutraceuticals and functional foods [14]; however, they are considered midway between food and pharmaceuticals. The best definition that I prefer is that nutraceuticals are bioactive constituents prepared from food and can treat or prevent one or more chronic diseases and sold in the form of pharmaceuticals meanwhile functional food is a food rich in bioactive ingredients and can prevent one or more chronic diseases and consumed in form of food. Our research team investigated the potential beneficial effect of nutraceuticals that could be efficient in the protection/or treatment of NASH through a series of researches (Figure 1).
Nutraceuticals composed of chitosan, and ferulic acid were shown to be efficient in treating steatohepatitis, the nano-form of such nutraceuticals showed superiority in this respect [15]. The mechanism was related to reducing insulin resistance, oxidative stress, inflammation (represented by high sensitivity C-reactive protein and adiponectin) liver triglycerides and total cholesterol.
Nigella sativa crude oil extracted by a screw press machine was shown to prevent inflammatory fatty liver and dyslipidemia in experimental rats and has antioxidant and anti-inflammatory activity [16]. It reduced Low-Density Lipoprotein-Cholesterol (LDL-C), Total Cholesterol (TC), and Triglycerides (TG) and elevated High-Density Lipoprotein-Cholesterol (HDL-C) together with reduction of malondialdehyde and tumor necrosis factor-α and improved liver function which was ascribed to the presence of 1% volatile constituents in the crude oil with major compounds represented by p-cymene and thymoquinone. Phytosterols especially β-sitosterol in addition to gamma and α-tocopherols in crude oil could also participate in the prevention of NASH [17,18].
Quinoa seeds were reported to be a good source of both lipophilic and hydrophilic nutraceuticals that could improve fatty liver due to the presence of α-tocopherols, linolenic fatty acids, phenolic compounds with protocatechuic, para hydroxybenzoic, ferulic and cinnamic acid as the major phenolic acid and rutin, chrysin and luteolin as the prominent flavonoids in two varieties of quinoa. Both nutraceuticals possess in-vitro and in-vivo antioxidant effects, with hypolipidemic, anti-inflammatory, and liver lipid-lowering effects with the improvement of liver and heart histopathology in rats. They produced a reduction in body weight gain and TC/HDL-C therefore reducing the risk of CVDs [19,20].
Clove essential oil conventional emulsion, eugenol nano-emulsion, the main constituent of the essential oil, and the nano-emulsion of the essential oil were studied in rats with dietary-induced fatty liver. The study showed that the three preparations improved fatty liver and dyslipidemia with protection from cardiovascular diseases and fatty liver complications [21].
The potential protective effect of Basil Essential Oil (BO) and its Nano-Emulsion (BNO) was studied towards NASH in rat models. Both treatments produced significant protection from NASH through a reduction in liver lipids, plasma TC, TG, LDL-C, TG/HDL-C, lipocaline, and interleukin-6. Microbiota represented by Firmicutes/Bacteriodetes (F/B) was improved. BNO was superior in reducing the F/B ratio, liver lipids, and liver histopathological changes while BO was more efficient in reducing TC, TG, and LDL-C. The therapeutic effect of BO and BNO was ascribed to the presence of linalool, eugenol, and eucalyptol as bioactive constituents [22].
Conclusion
Steatohepatitis is a serious condition that is commonly present in diabetics and obese subjects. Steatohepatitis is accused of developing cardiovascular diseases and might progress to hepatocellular carcinoma and liver failure. Experimental studies showed efficient nutraceuticals for the protection and treatment of NASH. The sources of such nutraceuticals are represented by Nigella sativa, quinoa, clove seed, and basil. Consumption of such sources in daily life could be beneficial in the prevention of steatohepatitis. Other nutraceuticals prepared from chitosan and ferulic acid showed efficiency in treating steatohepatitis and prevention of its complications. Prospective clinical studies are needed for confirmation of the efficiency of such nutraceuticals in humans.
- El-Attar MM, El-Melegy NT. Serum levels of leptin and adiponectin in patients with nonalcoholic fatty liver disease: potential biomarkers. JASMR. 2010; 5(2): 101–108.
- Al-Okbi SY. Role of nutraceuticals in prevention of non-alcoholic fatty liver. in Plant- and marine-based phytochemicals for human health: Attributes, potential, and use (Goyal MR; Chauhan DN. ed). Apple Academic Press, Taylor and Francis. 2019; 131-149.
- Xu X, Poulsen KL, Wu L, Liu S, Miyata T, Song Q, Wei Q, Zhao C, Lin C, Yang J. Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH). Signal Transduct Target Ther. 2022 Aug 13;7(1):287. doi: 10.1038/s41392-022-01119-3. PMID: 35963848; PMCID: PMC9376100.
- Kanwal F, Shubrook JH, Younossi Z, Natarajan Y, Bugianesi E, Rinella ME, Harrison SA, Mantzoros C, Pfotenhauer K, Klein S, Eckel RH, Kruger D, El-Serag H, Cusi K. Preparing for the NASH Epidemic: A Call to Action. Gastroenterology. 2021; 161(3):1030-1042.e8. doi: 10.1053/j.gastro.2021.04.074
- Fraile JM, Palliyil S, Barelle C, Porter AJ, Kovaleva M. Non-Alcoholic Steatohepatitis (NASH) - A Review of a Crowded Clinical Landscape, Driven by a Complex Disease. Drug Des Devel Ther. 2021 Sep 22;15:3997-4009. doi: 10.2147/DDDT.S315724. PMID: 34588764; PMCID: PMC8473845.
- Al-Okbi SY, Abd El Ghani S, Elbakry HFH, Mabrok HB, Nasr SM, Desouky HM. Kishk Sa′eedi as a potential functional food for management of metabolic syndrome: A study of the possible interaction with pomegranate seed oil and/ or gum Arabic. J Herbmed Pharmacol. 2021; 10(3): 319-330. doi: 10.34172/jhp.2021.37
- Sattar N, Forrest E, Preiss D. Non-alcoholic fatty liver disease. BMJ. 2014 Jul 29;349:g4596. doi: 10.1136/bmj.g4596. PMID: 25239614; PMCID: PMC4168663.
- Lee JH, Lee JJ, Cho WK, Yim NH, Kim HK, Yun B, Ma JY. KBH-1, an herbal composition, improves hepatic steatosis and leptin resistance in high-fat diet-induced obese rats. BMC Complement Altern Med. 2016 Sep 13;16(1):355. doi: 10.1186/s12906-016-1265-z. PMID: 27618865; PMCID: PMC5020448.
- Aygun C, Senturk O, Hulagu S, Uraz S, Celebi A, Konduk T, Mutlu B, Canturk Z. Serum levels of hepatoprotective peptide adiponectin in non-alcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2006 Feb;18(2):175-80. doi: 10.1097/00042737-200602000-00010. PMID: 16394799.
- Bashiardes S, Zilberman-Schapira G, Elinav E. Use of Metatranscriptomics in Microbiome Research. Bioinform Biol Insights. 2016 Apr 20;10:19-25. doi: 10.4137/BBI.S34610. PMID: 27127406; PMCID: PMC4839964.
- Raza S, Rajak S, Upadhyay A, Tewari A, Anthony Sinha R. Current treatment paradigms and emerging therapies for NAFLD/NASH. Front Biosci (Landmark Ed). 2021 Jan 1;26(2):206-237. doi: 10.2741/4892. PMID: 33049668; PMCID: PMC7116261.
- Harrison SA, Allen AM, Dubourg J, Noureddin M, Alkhouri N. Challenges and opportunities in NASH drug development. Nat Med. 2023 Mar;29(3):562-573. doi: 10.1038/s41591-023-02242-6. Epub 2023 Mar 9. PMID: 36894650.
- Matafome P, Louro T, Rodrigues L, Crisóstomo J, Nunes E, Amaral C, Monteiro P, Cipriano A, Seiça R. Metformin and atorvastatin combination further protect the liver in type 2 diabetes with hyperlipidaemia. Diabetes Metab Res Rev. 2011 Jan;27(1):54-62. doi: 10.1002/dmrr.1157. PMID: 21218508.
- Aronson JK. Defining 'nutraceuticals': neither nutritious nor pharmaceutical. Br J Clin Pharmacol. 2017 Jan;83(1):8-19. doi: 10.1111/bcp.12935. Epub 2016 Apr 25. PMID: 26991455; PMCID: PMC5338166.
- Al-Okbi SY, Ali O, Aly AS, Refaat D, Esmail RSH, Elbakry HFH. Management of metabolic syndrome by nutraceuticals prepared from chitosan and ferulic acid with or without beta-sitosterol and their nanoforms. Sci Rep. 2023 Jul 27;13(1):12176. doi: 10.1038/s41598-023-38837-9. PMID: 37500657; PMCID: PMC10374579.
- Al-Okbi SY, Mohamed DA, Hamed TE, Edris AE. Potential protective effect of Nigella sativa crude oils towards fatty liver in rats. Eur J Lipid Sci Technol. 2013; 115: 774-782.
- Cheikh-Rouhou S, Besbes S, Lognay G, Blecker C. Sterol composition of black cumin (Nigellas ativa L.) and Aleppo pine (Pinus halepensis Mill.) seed oils. J Food Comp Anal. 2008; 21: 162-168.
- Ramadan F, Wahdan K, Blending of corn oil with black cumin (Nigella sativa) and coriander (Coriandrum sativum) seed oils: Impact on functionality, stability and radical scavenging activity. Food Chem. 2012; 132: 873–879.
- Al-Okbi SY, Hamed TE, Elewa TA, Ramadan AA, Bakry BA, El Karamany MF. Quinoa seed: A source of lipophilic nutraceuticals for prevention of metabolic syndrome in rat model. Grasas Aceites. 2024; e542. https://doi.org/10.3989/gya.1104222
- Al-Okbi SY, Hamed TE, Elewa TA, Ramadan AA, El-Karamany MF, Bakry BA. Role of polar extracts from two quinoa varieties in prevention of steatohepatitis and cardiovascular diseases and improving glucose tolerance in rats. J Herbmed Pharmacol.2021; 10(1): 93-101. doi: 10.34172/jhp.2021.09.
- Al-Okbi SY, Mohamed DA, Hamed TE and Edris AE. Protective Effect of Clove Oil and Eugenol Microemulsions on Fatty Liver and Dyslipidemia as Components of Metabolic Syndrome. J Medicinal Food. 2014; 17(7): 764-771. DOI: 10.1089/jmf.2013.0033
- Al-Okbi SY, Amin MA, Mohamed AEA, Edris AE, Sharaf OM, Mabrok HB, Ramadan AA. Basil Essential Oil and Its Nanoemulsion Mitigate Non Alcoholic Steatohepatitis in Rat Model with Special Reference to Gut Microbiota. J Oleo Sci. 2020 Aug 6;69(8):913-927. doi: 10.5650/jos.ess20067. Epub 2020 Jul 9. PMID: 32641615.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
